Cell biology of apicomplexan parasites
HEADS OF THE TEAM
Dr. Maryse Lebrun & Dr. Sébastien Besteiro


RESEARCH INTERESTS
We are interested in the cellular and molecular mechanisms that enable apicomplexan parasites to infect and develop inside host. Apicomplexan are single-celled eukaryotes parasites. They are important pathogens of humans and domestic animals. They include Plasmodium spp. the causative agent of malaria. Malaria is responsible for almost half million human deaths per year and increasing drug-resistance is a considerable concern. It also includes Toxoplasma, a prominent cause of human congenital infections and Cryptosporidium, a leading cause of severe diarrhea and mortality in infants. No vaccine currently exists against these parasites.
The objectives of our team are to decipher cellular and molecular features related to two essential parasitic functions: host cell invasion and intracellular replication, with the aim of identifying novel therapeutic strategies. Our research is mainly focused on T. gondii, which is a relevant model for many features conserved throughout the phylum. We then extend our most important discoveries to the malaria parasite. We use a broad array of cell biological and biochemical approaches, as well as state-of the art molecular genetics and genome editing.
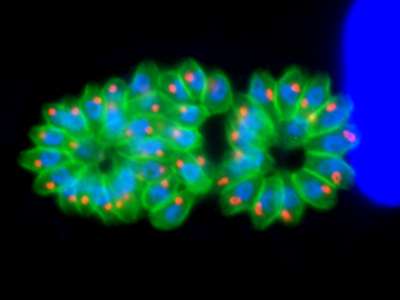
The team is currently composed of 3 research groups:
- Group 1 – Host cell invasion and rhoptry secretion (PI Maryse Lebrun)
- Group 2 – Role of the apicoplast in T. gondii metabolism and survival (PI Sébastien Besteiro)
- Group 3 – Egress and protein phosphatases in P. falciparum (PI Mauld Lamarque)
NEWS
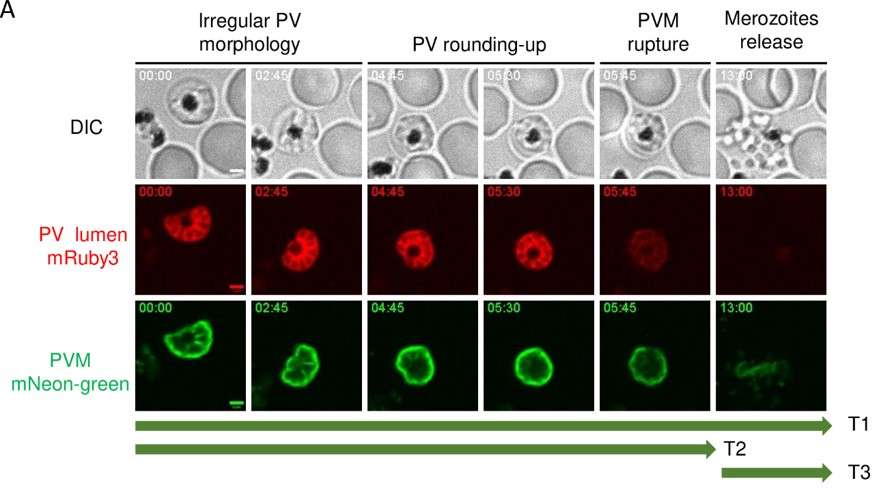
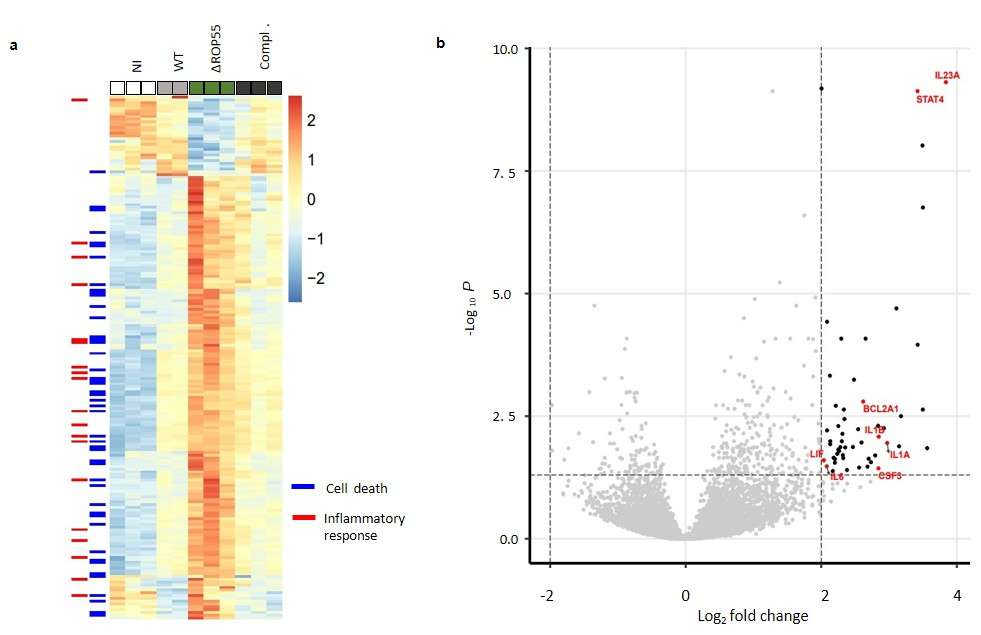
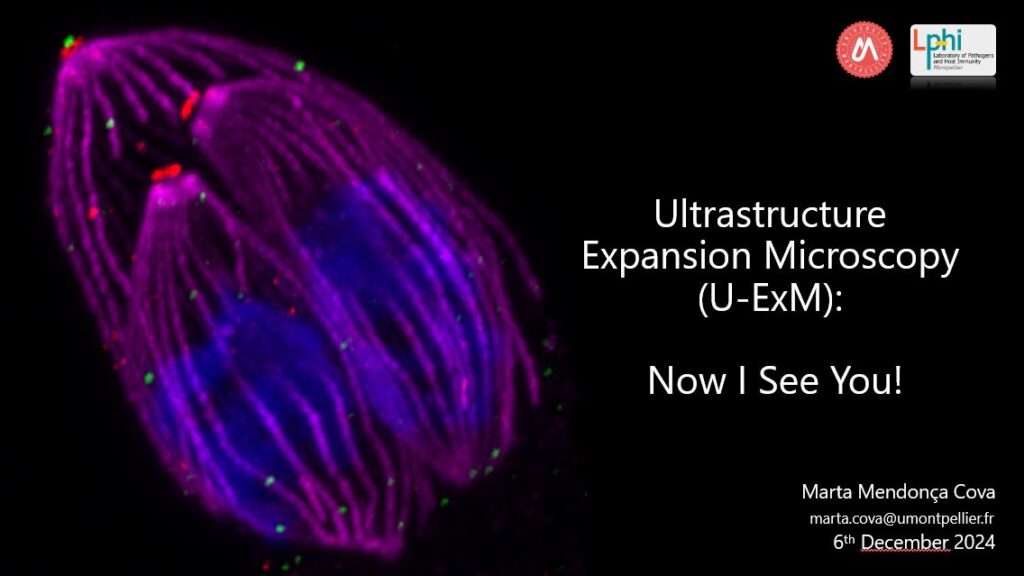
FUNDING
Our team has received a team label from ‘Fondation pour la Recherche Médicale’ (‘Medical Research Foundation’ – FRM) both in 2013 and in 2016.
Maryse Lebrun has been been awarded the ERC Advanced grant 2018 over 5 years by the European Council to unravel the mechanism of eukaryote secretion system.
Past and current funding:
